Description
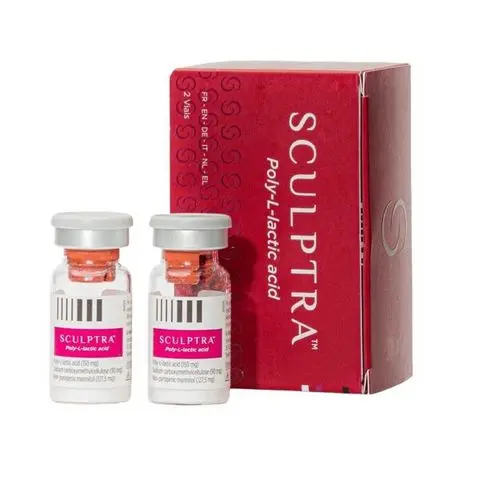
What is the sculptra poly lactic-acid (PLLA)?
Injectable poly-l-lactic acid (PLLA) is known commercially as Sculptra and Sculptra Aesthetic. The FDA has approved Sculptra since 2004 to treat lipoatrophy (thinning of the fat pads on the face) associated with the HIV virus. Sculptra Aesthetic received its FDA approval in 2009 for improving the appearance of nasolabial folds contour changes and other facial wrinkles.
As we age, the fat, muscles, bone, and skin in our face begins to thin. This loss of volume leads to either a sunken or sagging appearance of the face. Injectable poly-l-lactic acid is used to create structure, framework, and volume to the face. PLLA is known as a bio-stimulatory dermal filler, this means it stimulates your own skin to produce new collagen. Over time your skin breaks down PLLA into water and carbon dioxide. The effects of PLLA appear gradually over a few months, producing natural results.
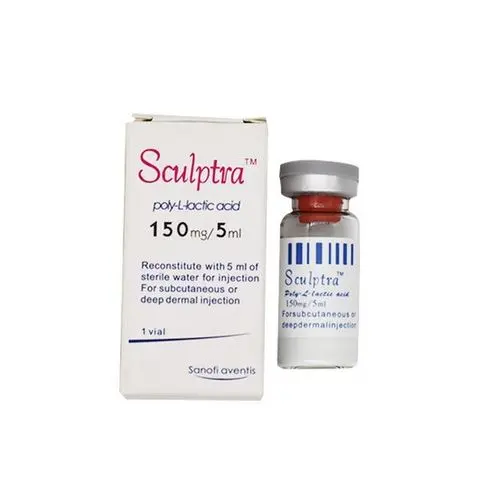
- Sculptra
- Sculptra PLLA
- sculptra poly-L-lactic acid
Production Capacity:
50000000
Delivery Timeframe:
Immediate
Incoterms:
DDU - Delivered Duty Unpaid
FOB - Free on Board
Packaging Details:
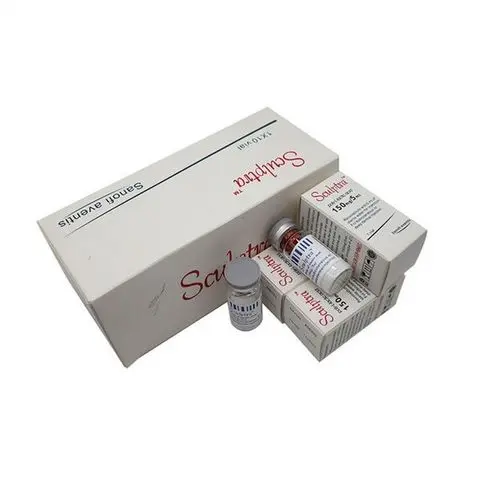
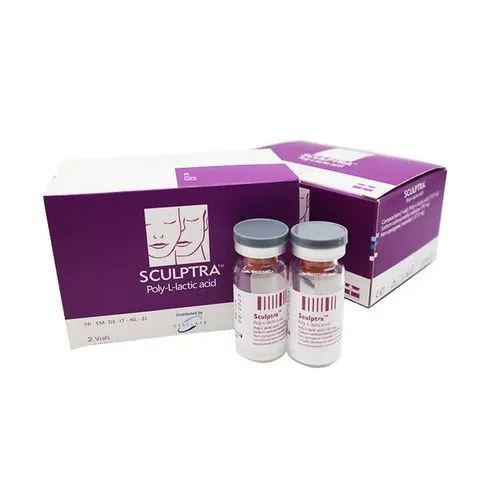
Sculptra : 150mg/5ml x 1 vail per box Scukptra PLLA/Gladerma : 150mg/5ml x 2 vails per box
More about
Nuro Beauty technology co.,ltd
10-50
Employees
500K - 1M
Sales volume (USD)
100%
% Export sales
Year
Established
Business type
- Industry / Manufacturer
- Representative / Agent
- Distributor / Wholesaler
Keywords
- 玻尿酸
- 蛋白线
- 美容产品
Contact and location
-
youki ********
-
+86 1********
-
shijiazhuang / | China